5.6 Blood Typing
Leanne Dooley
Learning Objectives
By the end of this section, you will be able to:
- Describe the two basic physiological consequences of transfusion of incompatible blood
- Compare and contrast ABO and Rh blood groups
- Identify which blood groups may be safely transfused into patients with different ABO types
- Discuss the pathophysiology of haemolytic disease of the newborn
Blood transfusions in humans were risky procedures until the discovery of the major human blood groups by Karl Landsteiner, an Austrian biologist and physician, in 1900. Until that point, physicians did not understand why rapid death often followed blood transfusions. They assumed that all human blood was the same. Landsteiner discovered that the plasma of some individuals caused the red blood cells of some other individuals to clump together (agglutinate), and he surmised that fatal transfusion reaction occurred when the type of donor blood infused into the patient was incompatible with the patient’s own blood. Blood groups are determined by the presence or absence of specific marker molecules on the plasma membranes of erythrocytes. With the discovery of these inherited markers, it became possible for the first time to match patient-donor blood types and prevent severe haemolytic transfusion reactions and deaths.
Antigens, Antibodies and Transfusion Reactions
Antigens are substances that the body does not recognise as belonging to the “self” and that therefore trigger a defensive response from the leukocytes of the immune system. (Seek more content for additional information on immunity.) Here, we will focus on the role of antibody-mediated (humoral) immunity in blood transfusion reactions. With RBCs in particular, you may see the antigens referred to as isoantigens or agglutinogens (surface antigens) and the antibodies referred to as isoantibodies or agglutinins. In this chapter, we will use the more common terms antigens and antibodies.
Antigens are generally large proteins, but may include other classes of organic molecules, including carbohydrates, lipids, and nucleic acids. Following an infusion of incompatible blood, erythrocytes with foreign antigens appear in the bloodstream and trigger an immune response. Proteins called antibodies (immunoglobulins), which are produced by certain B lymphocytes called plasma cells, attach to the antigens on the plasma membranes of the infused erythrocytes. Large antibodies, called IgM antibodies, can cause direct destruction of incompatible RBC via activation of group of plasma proteins called complement. This type of RBC destruction is called intravascular haemolysis and the reaction is referred to as an acute haemolytic transfusion reaction, which can be fatal after infusion of as little as 10mL of incompatible RBC. Smaller antibodies, called IgG antibodies, bind to the incompatible RBC and mark them for destruction by macrophages in the spleen. This type of RBC destruction is called extravascular haemolysis and the reaction is called a delayed haemolytic transfusion reaction, which is not often fatal but leads to removal of the transfused RBC, effectively making the transfusion a waste of time.
During an acute haemolytic transfusion reaction haemoglobin is released into the bloodstream as a result of intravascular haemolysis. This haemoglobin travels to the kidneys, which are responsible for filtration of the blood. However, haemoglobin is toxic to endothelial cells in the kidney and the patient can quickly develop kidney failure. In addition, damage to these endothelial cells can trigger coagulation and acute inflammatory response that can lead to shock and death within minutes.
More than 50 antigens have been identified on erythrocyte membranes, but by far the most significant in terms of their potential harm to patients are the A and B antigens of the ABO blood group system discovered by Landsteiner. The next most clinically significant antigen is the D antigen of the Rhesus or Rh blood group system.
The ABO Blood Group
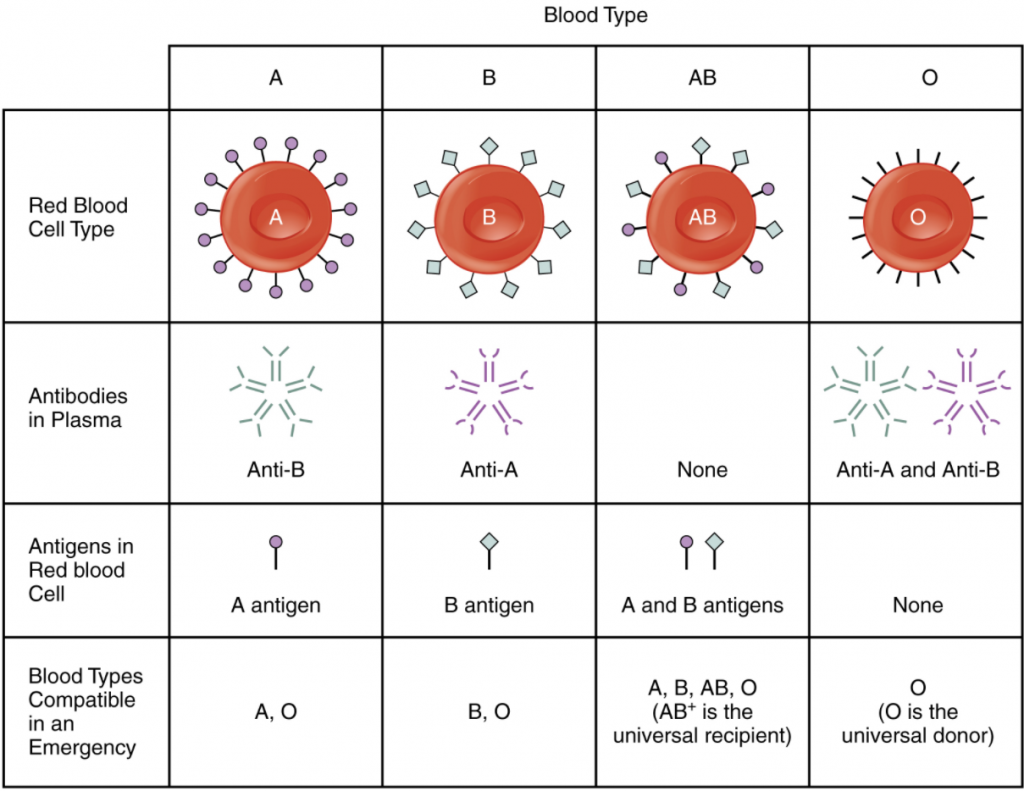
Although the ABO blood group name consists of three letters, ABO blood typing designates the presence or absence of just two antigens, A and B. Both are carbohydrate or saccharide antigens and their presence or absence on the RBC membrane is genetically determined. People who have A antigens only on their erythrocyte membrane surfaces are designated blood type A, and those whose erythrocytes have B antigens only are blood type B. People can also have both A and B antigens on their erythrocytes, in which case they are blood type AB. People with neither A nor B antigens are designated blood type O.
Normally the body must be exposed to a foreign antigen before an antibody can be produced. This is not the case for the ABO blood group. Individuals with type A blood—without any prior exposure to incompatible blood—have pre-formed or naturally occurring antibodies to the B antigen circulating in their blood plasma. These antibodies, referred to as anti-B antibodies, are IgM type antibodies and will cause agglutination and haemolysis if they ever encounter erythrocytes with B antigens. Similarly, an individual with type B blood has pre-formed anti-A antibodies. Individuals with type AB blood, which has both antigens, do not have pre-formed antibodies to either of these. People with type O blood lack antigens A and B on their erythrocytes, but both anti-A and anti-B antibodies circulate in their blood plasma. Anti-A and anti-B antibodies are not present at birth but develop during the first six months of life in response to A and B-like substances in the environment, especially of the surface of bacteria.
Rh Blood Groups
The Rhesus or Rh blood group is classified according to the presence or absence of a second erythrocyte antigen identified as Rh. (It was first discovered in a type of primate known as a rhesus macaque, which is often used in research, because its blood is like that of humans.) Although dozens of Rh antigens have been identified, the antigen, designated D, is the most clinically important. Those who have the Rh D antigen present on their erythrocytes—about 85 percent of Americans—are described as Rh positive (Rh+) and those who lack it are Rh negative (Rh−). Note that the Rh group is distinct from the ABO group, so any individual, no matter their ABO blood type, may or may not have Rh antigen on their RBCs. When identifying a patient’s blood type, the Rh group is designated by adding the word positive or negative to the ABO type. For example, A positive (A+) means ABO group A blood with the Rh(D) antigen present, and AB negative (AB−) means ABO group AB blood without the Rh(D) antigen.
In contrast to the ABO group antibodies, which are preformed, antibodies to the Rh antigen are produced only in Rh− individuals after exposure to the antigen. This process, called sensitisation, occurs following a transfusion with Rh-incompatible blood or, more commonly, with the birth of an Rh+ baby to an Rh− mother. Problems are rare in a first pregnancy, since the baby’s Rh+ cells rarely cross the placenta (the organ of gas and nutrient exchange between the baby and the mother). However, during or immediately after birth, the Rh− mother can be exposed to the baby’s Rh+ cells (Figure 5.6.1). Research has shown that this occurs in about 13−14 percent of such pregnancies. After exposure, the mother’s immune system begins to generate anti-Rh (anti-D) antibodies. If the mother should then conceive another Rh+ baby, the Rh antibodies she has produced, which are small IgG antibodies, can cross the placenta into the foetal bloodstream and mark the foetal RBCs fir destruction by macrophages in the foetal spleen. This condition, known as haemolytic disease of the foetus and newborn (HDFN) or erythroblastosis foetalis, may cause anaemia in mild cases, but the haemolysis can be so severe that without treatment the foetus may die in the uterus or shortly after birth. An Rh- mother can also produce Rh antibodies after a miscarriage, termination of pregnancy or abdominal trauma during pregnancy with an Rh+ foetus.
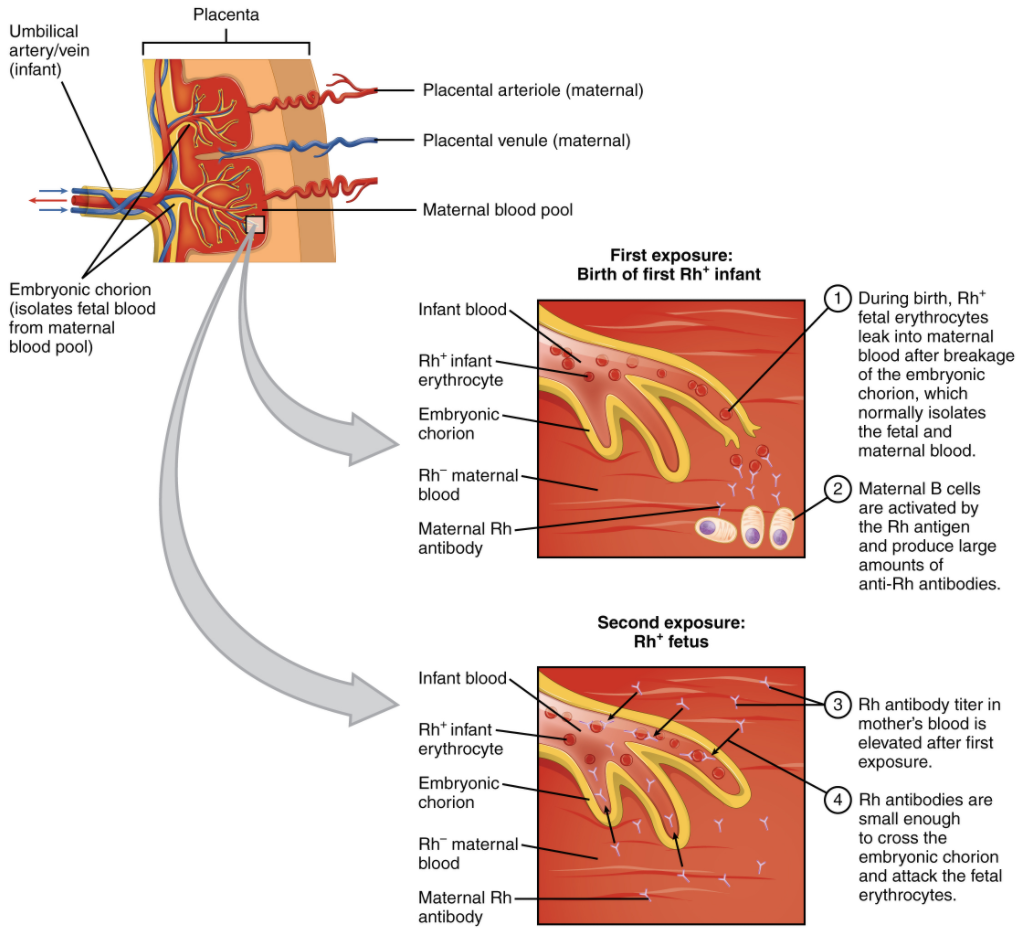
Rh (D) immune globulin (Rh(D)Ig), derived from the plasma of women who have developed Rh antibodies, can temporarily prevent the development of Rh antibodies in the Rh− mother, thereby averting this potentially serious disease for the foetus. The anti-Rh antibodies, injected into the Rh- mother destroy any foetal Rh+ erythrocytes that may cross the placental barrier and enter the mother’s circulation. This effectively prevents sensitisation and the mother’s immune system does not begin to produce Rh antibodies. Rh(D)Ig is normally administered to Rh− mothers during weeks 26−28 of pregnancy, within 72 hours following birth of an Rh+ baby and following any potentially sensitive event. It has proven remarkably effective in decreasing the incidence of HDFN. Earlier we noted that the incidence of HDFN in an Rh+ subsequent pregnancy to an Rh− mother is about 13–14 percent without preventive treatment.
Determining ABO Blood Types
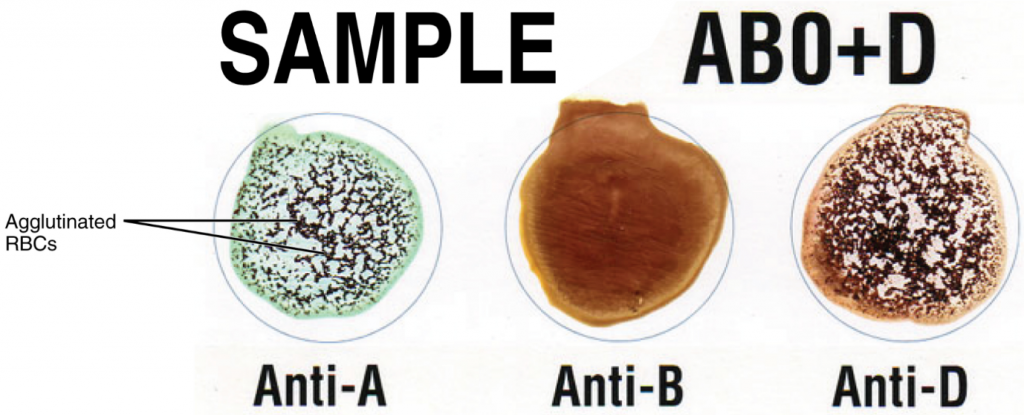
Laboratory technicians and scientists are able to determine a patient’s blood type quickly and easily using commercially prepared antibodies. A drop of the patient’s blood sample is placed into separate wells on a plastic tile or into separate glass test tubes. Into one well a drop of commercially prepared anti-A antibody is added and to another a drop of anti-B antibody is added. If the antigen is present, the antibodies will cause visible agglutination of the cells (Figure 5.6.2). The patient’s blood should also be tested for the Rh(D) antigen using commercially prepared anti-Rh(D) antibodies.
ABO Transfusion Protocols
To avoid transfusion reactions, it is best to transfuse only matching blood types; that is, a type B+ recipient should ideally receive blood only from a type B+ donor and so on. That said, in emergency situations, when acute haemorrhage threatens the patient’s life, there may not be time for blood typing and cross matching. In these cases, blood from a universal RBC donor—an individual with type O− blood—may be transfused. Recall that type O erythrocytes do not display A or B antigens. Thus, anti-A or anti-B antibodies that might be circulating in the patient’s blood plasma will not encounter any erythrocyte surface antigens on the donated blood and therefore will not be provoked into a response. A patient with blood type AB+ is known as the universal RBC recipient. This patient can theoretically receive RBC of any type of blood, because the patient’s own plasma – having both A and B antigens on the erythrocyte surface – does not contain anti-A or anti-B antibodies. Figure 5.6.3 summarises the ABO blood types and their associated antigen and antibody profiles.
Note that type A RBC can be given to recipients with type A or type AB blood, because both cases, the recipient’s plasma does not contain anti-A antibodies. However, type AB RBC cannot be given to recipients with type A blood because in this case the patient’s plasma will contain anti-B antibodies, which will react with the B antigens on the donor blood.
While most transfusions involve infusion of RBC only, some patients require plasma transfusions. In the case of plasma transfusion, type AB is the universal donor because type AB plasma does not contain either anti-A or anti-B. The Rh(D) antigen also plays a role in blood transfusion. If Rh− individuals receiving blood have had prior exposure to Rh antigen, antibodies for this antigen may be present in the plasma and will trigger delayed haemolytic transfusion reaction and extravascular haemolysis. Although it is always preferable to type and cross match a patient’s blood before transfusing, in a true life-threatening emergency, this is not always possible, and type O- RBC or type AB plasma can be given until the patient’s blood type is established.
At the scene of multiple-vehicle accidents, military engagements, and natural or human-caused disasters, many victims may suffer simultaneously from acute haemorrhage, yet type O blood may not be immediately available. In these circumstances, medics may at least try to replace some of the volume of blood that has been lost. This is done by intravenous administration of a saline solution that provides fluids and electrolytes in proportions equivalent to those of normal blood plasma. Research is ongoing to develop a safe and effective artificial blood that would perform the oxygen-carrying function of blood without the RBCs, enabling transfusions in the field without concern for incompatibility.
Section Review
Antigens are non-self molecules, usually large proteins, which provoke an immune response. In transfusion reactions, antibodies attach to antigens on the surfaces of erythrocytes and cause agglutination and haemolysis. ABO blood group antigens are designated A and B. People with type A blood have A antigens on their erythrocytes, whereas those with type B blood have B antigens. Those with AB blood have both A and B antigens, and those with type O blood have neither A nor B antigens. The blood plasma contains preformed antibodies against the antigens not present on a person’s erythrocytes.
A second group of clinically important blood antigens is the Rh group, the most important of which is Rh D. People with Rh− blood do not have this antigen on their erythrocytes, whereas those who are Rh+ do. About 85 percent of Americans are Rh+. When a woman who is Rh− becomes pregnant with an Rh+ foetus, her body may begin to produce anti-Rh antibodies. If she subsequently becomes pregnant with a second Rh+ foetus and has not been treated preventively with Rh(D)Ig, the foetus will be at risk for haemolytic disease of the foetus and newborn.
Agglutination testing to determine blood type is necessary before transfusing blood, unless the patient is experiencing haemorrhage that is an immediate threat to life, in which case type O− red blood cells or type AB plasma may be transfused.
Review Questions
Critical Thinking Questions
Click the drop down below to review the terms learned from this chapter.

