17.5 Physiology of Urine Formation
Learning Objectives
By the end of this section, you will be able to:
- Describe the hydrostatic and colloid osmotic forces that favour and oppose filtration
- Describe glomerular filtration rate (GFR), state the average value of GFR and explain how clearance rate can be used to measure GFR
- Predict specific factors that will increase or decrease GFR
- State the percent of the filtrate that is normally reabsorbed and explain why the process of reabsorption is so important
- Calculate daily urine production
- List common symptoms of kidney failure
Having reviewed the anatomy and microanatomy of the urinary system, now is the time to focus on the physiology. You will discover that different parts of the nephron utilise specific processes to produce urine: filtration, reabsorption and secretion. You will learn how each of these processes works and where they occur along the nephron and collecting ducts. The physiologic goal is to modify the composition of the plasma and, in doing so, produce the waste product urine.
Failure of the renal anatomy and/or physiology can lead suddenly or gradually to renal failure. In this event, a number of symptoms, signs, or laboratory findings point to the diagnosis (Table 17.5.1).
Table 17.5.1. Symptoms of Kidney Failure
| Symptoms | ||
| Weakness | Lethargy | Shortness of breath |
| Widespread oedema | Anaemia | Metabolic acidosis |
| Metabolic alkalosis | Heart arrhythmias | Uraemia (high urea level in the blood) |
| Loss of appetite | Fatigue | Excessive urination |
| Oliguria (too little urine output) |
Glomerular Filtration Rate (GFR)
The volume of filtrate formed by both kidneys per minute is termed the glomerular filtration rate (GFR). The heart pumps about 5 L blood per min under resting conditions. Approximately 20-25 percent or one to 1.5 litres enters the kidneys to be filtered. On average, this results in the production of about 125 mL/min filtrate produced in men (range of 90 to 140 mL/min) and 105 mL/min filtrate produced in women (range of 80 to 125 mL/min). This then equates to a volume of about 180 L/day in men and 150 L/day in women. Ninety-nine percent of this filtrate is returned to the circulation by reabsorption so that only about 1–2 litres of urine are produced per day (Table 17.5.2).
Table 17.5.2. Calculating urine formation per day
| Flow | Flow per minute (mL) | Calculation |
| Renal blood flow | 1050 | Cardiac output is about 5000 mL/minute, of which 21% flows through the kidney
5000*0.21 = 1050 mL blood/min |
| Renal plasma flow | 578 | Renal plasma flow equals to the blood flow per minute times the haematocrit. If a person has a haematocrit of 45, then the renal plasma flow is 55%.
1050*0.55 = 578 mL plasma/min |
| Glomerular filtration rate | 110 | The GFR is the amount of plasma entering Bowman’s capsule per minute. It is the renal plasma flow times the filtration that enters the renal capsule (19%).
587*0.55 = 110 mL filtrate/min |
| Urine | 1296 | The filtrate not recovered by the kidney is the urine that will be eliminated. It is the GFR times the fraction of the filtrate that is not reabsorbed (0.8%).
110*0.008 = 0.9 mL urine/min
Multiply urine/min times 60 minutes times 24 hours to get daily urine production. 0.9*60*24 = 1296 mL/day urine |
GFR is influenced by the hydrostatic pressure and colloid osmotic pressure on either side of the capillary membrane of the glomerulus. Recall that filtration occurs as pressure forces fluid and solutes through a semipermeable barrier with the solute movement constrained by particle size. Hydrostatic pressure is the pressure produced by a fluid against a surface. If you have a fluid on both sides of a barrier, both fluids exert a pressure in opposing directions. Net fluid movement will be in the direction of the lower pressure. Osmosis is the movement of solvent (water) across a membrane that is impermeable to a solute in the solution. This creates a pressure, osmotic pressure, which will exist until the solute concentration is the same on both sides of a semipermeable membrane. If the concentration differs, water will move. Glomerular filtration occurs when glomerular hydrostatic pressure exceeds the luminal hydrostatic pressure of Bowman’s capsule. There is also an opposing force, the osmotic pressure, which is typically higher in the glomerular capillary.
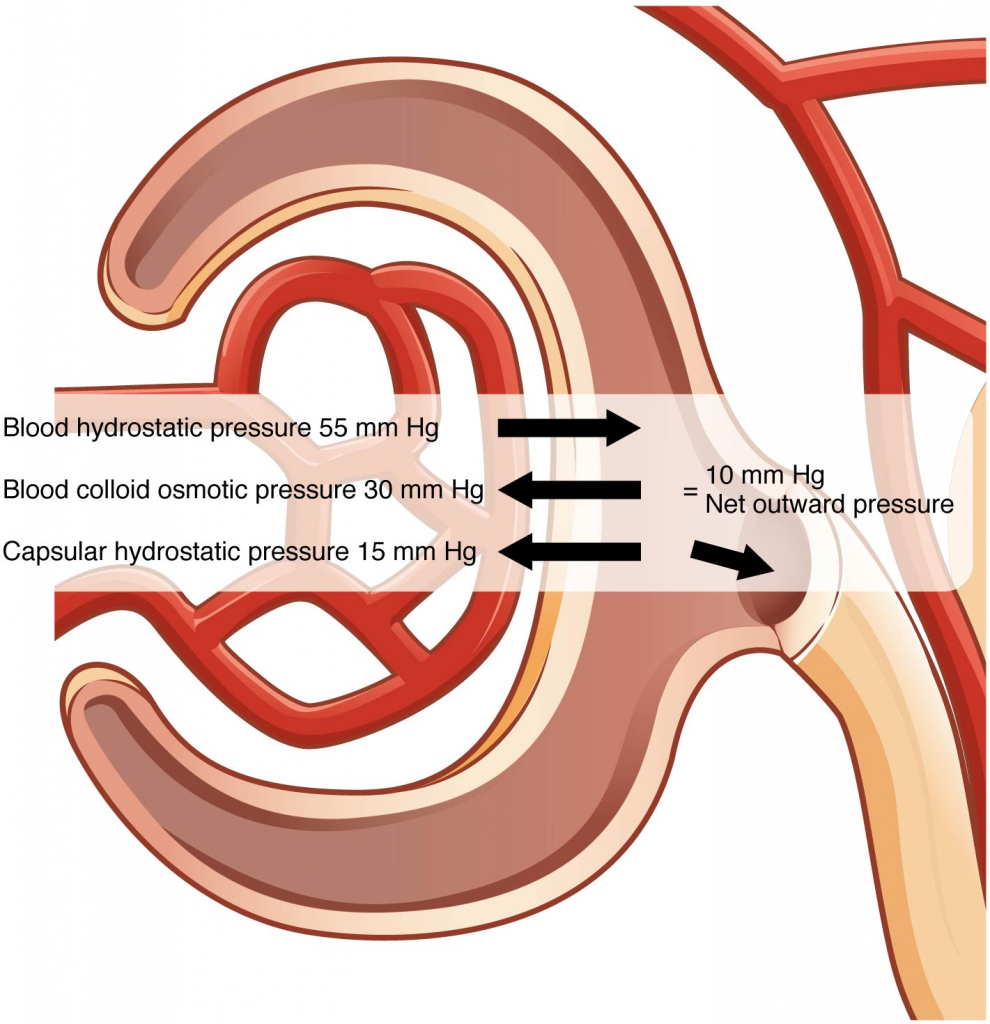
To understand why this is so, look more closely at the microenvironment on either side of the filtration membrane. You will find osmotic pressure exerted by the solutes inside the lumen of the capillary as well as inside of Bowman’s capsule. Since the filtration membrane limits the size of particles crossing the membrane, the osmotic pressure inside the glomerular capillary is higher than the osmotic pressure in Bowman’s capsule. Recall that cells and the medium-to-large proteins cannot pass between the podocyte processes or through the fenestrations of the capillary endothelial cells. This means that red and white blood cells, platelets, albumins, and other proteins too large to pass through the filter remain in the capillary, creating an average colloid osmotic pressure of 30 mm Hg within the capillary. The absence of proteins in Bowman’s space (the lumen within Bowman’s capsule) results in an osmotic pressure near zero. Thus, the only pressure moving fluid across the capillary wall into the lumen of Bowman’s space is hydrostatic pressure. Hydrostatic (fluid) pressure is sufficient to push water through the membrane despite the osmotic pressure working against it. The sum of all the influences, both osmotic and hydrostatic, results in a net filtration pressure (NFP) of about 10 mm Hg (Figure 17.5.1).
A proper concentration of solutes in the blood is important in maintaining osmotic pressure both in the glomerulus and systemically. There are disorders in which too much protein passes through the filtration slits into the kidney filtrate. This excess protein in the filtrate leads to a deficiency of circulating plasma proteins. In turn, the presence of protein in the urine increases its osmolarity; this holds more water in the filtrate and results in an increase in urine volume. Because there is less circulating protein, principally albumin, the osmotic pressure of the blood falls. Less osmotic pressure pulling water into the capillaries tips the balance towards hydrostatic pressure, which tends to push it out of the capillaries. The net effect is that water is lost from the circulation to interstitial tissues and cells. This “plumps up” the tissues and cells, a condition termed systemic oedema.
Net Filtration Pressure (NFP)
NFP determines filtration rates through the kidney. It is determined as follows:
NFP = Glomerular blood hydrostatic pressure (GBHP) – [capsular hydrostatic pressure (CHP) + blood colloid osmotic pressure (BCOP)] = 10 mm Hg
That is:
NFP = GBHP – [CHP + BCOP] = 10 mm Hg
Or:
NFP = 55 – [15 + 30] = 10 mm Hg
As you can see, there is a low net pressure across the filtration membrane. Intuitively, you should realise that minor changes in osmolarity of the blood or changes in capillary blood pressure result in major changes in the amount of filtrate formed at any given point in time. The kidney can cope with a wide range of blood pressures. In large part, this is due to the autoregulatory nature of smooth muscle. When you stretch it, it contracts. Thus, when blood pressure goes up, smooth muscle in the afferent capillaries contracts to limit any increase in blood flow and filtration rate. When blood pressure drops, the same capillaries relax to maintain blood flow and filtration rate. The net result is a steady flow of blood into the glomerulus and a steady filtration rate despite significant systemic blood pressure changes. Mean arterial blood pressure is calculated by adding 1/3 of the difference between the systolic and diastolic pressures to the diastolic pressure. Therefore, if the blood pressure is 110/80, the difference between systolic and diastolic pressure is 30. One third of this is 10, and when you add this to the diastolic pressure of 80, you arrive at a calculated mean arterial pressure of 90 mm Hg. Therefore, if you use mean arterial pressure for the GBHP in the formula for calculating NFP, you can determine that if mean arterial pressure is above approximately 60 mm Hg, the pressure will be adequate to maintain glomerular filtration. Blood pressures below this level will impair renal function and cause systemic disorders that are severe enough to threaten survival. This condition is called shock.
Determination of the GFR is one of the tools used to assess the kidney’s excretory function. This is more than just an academic exercise. Since many drugs are excreted in the urine, a decline in renal function can lead to toxic accumulations. Additionally, administration of appropriate drug dosages for those drugs primarily excreted by the kidney requires an accurate assessment of GFR. GFR can be estimated closely by intravenous administration of inulin. Inulin is a plant polysaccharide that is neither reabsorbed nor secreted by the kidney. Its appearance in the urine is directly proportional to the rate at which it is filtered by the renal corpuscle. However, since measuring inulin clearance is cumbersome in the clinical setting, most often, the GFR is estimated by measuring naturally occurring creatinine, a protein-derived molecule produced by muscle metabolism that is not reabsorbed and only slightly secreted by the nephron.
Section Review
The entire volume of the blood is filtered through the kidneys about 300 times per day and 99 percent of the water filtered is recovered. The GFR is influenced by hydrostatic pressure and colloid osmotic pressure. Under normal circumstances, hydrostatic pressure is significantly greater and filtration occurs. The hydrostatic pressure of the glomerulus depends on systemic blood pressure, autoregulatory mechanisms, sympathetic nervous activity and paracrine hormones. The kidney can function normally under a wide range of blood pressures due to the autoregulatory nature of smooth muscle.
Review Questions
Review Questions
Click the drop down below to review the terms learned from this chapter.

