14.3 The Pituitary Gland and Hypothalamus
Learning Objectives
By the end of this section, you will be able to:
- Explain the interrelationships of the anatomy and functions of the hypothalamus and the posterior and anterior lobes of the pituitary gland
- Identify the two hormones released from the posterior pituitary, their target cells and their principal actions
- Identify the six hormones produced by the anterior lobe of the pituitary gland, their target cells, their principal actions and their regulation by the hypothalamus
The hypothalamus–pituitary complex can be thought of as the “command centre” of the endocrine system. This complex secretes several hormones that directly produce responses in target tissues, as well as hormones that regulate the synthesis and secretion of hormones of other glands. In addition, the hypothalamus–pituitary complex coordinates the messages of the endocrine and nervous systems. In many cases, a stimulus received by the nervous system must pass through the hypothalamus–pituitary complex to be translated into hormones that can initiate a response.
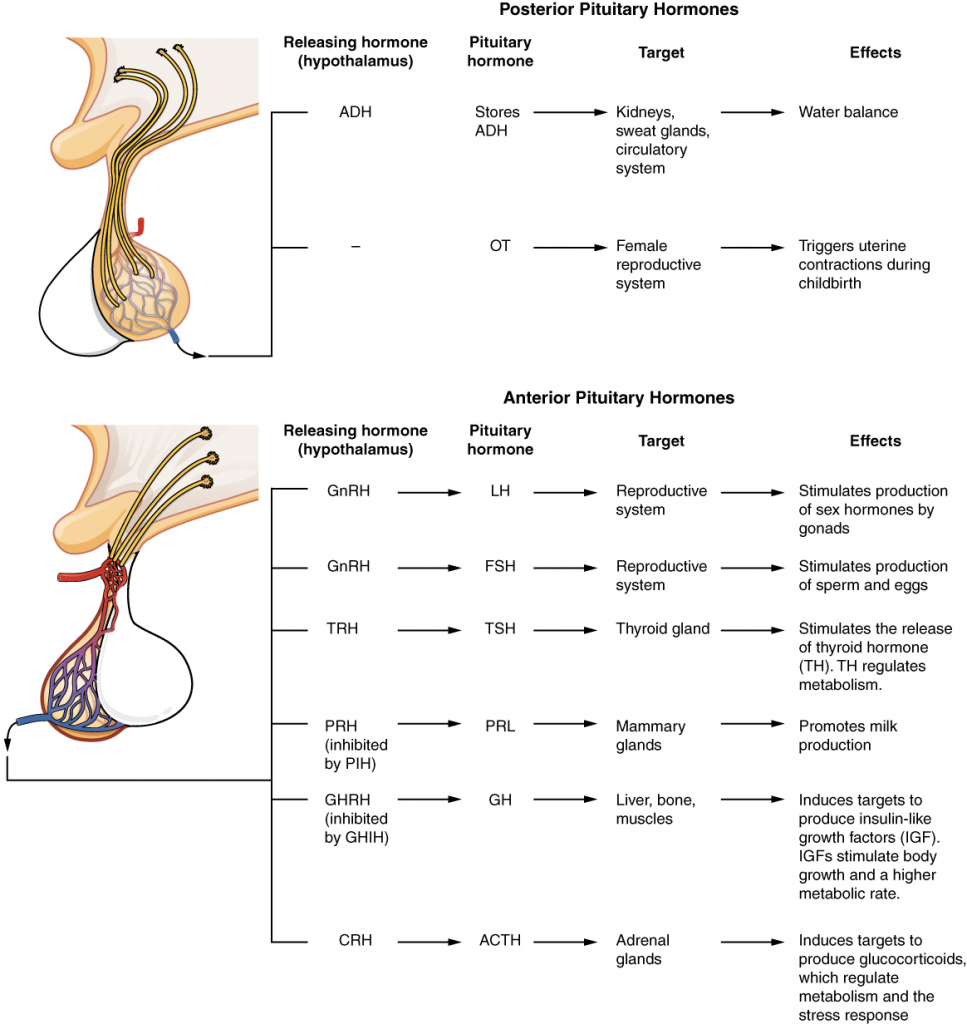
The hypothalamus is a structure of the diencephalon of the brain located anterior and inferior to the thalamus (Figure 14.3.1). It has both neural and endocrine functions, producing and secreting many hormones. In addition, the hypothalamus is anatomically and functionally related to the pituitary gland (or hypophysis), a bean-sized organ suspended from it by a stem called the infundibulum (or pituitary stalk). The pituitary gland is cradled within the sella turcica of the sphenoid bone of the skull. It consists of two lobes that arise from distinct parts of embryonic tissue: the posterior pituitary (neurohypophysis) is neural tissue, whereas the anterior pituitary (also known as the adenohypophysis) is glandular tissue that develops from the primitive digestive tract, specifically the developing hard palate. The hormones secreted by the posterior and anterior pituitary, and the intermediate zone between the lobes are summarised in Table 14.3.1.
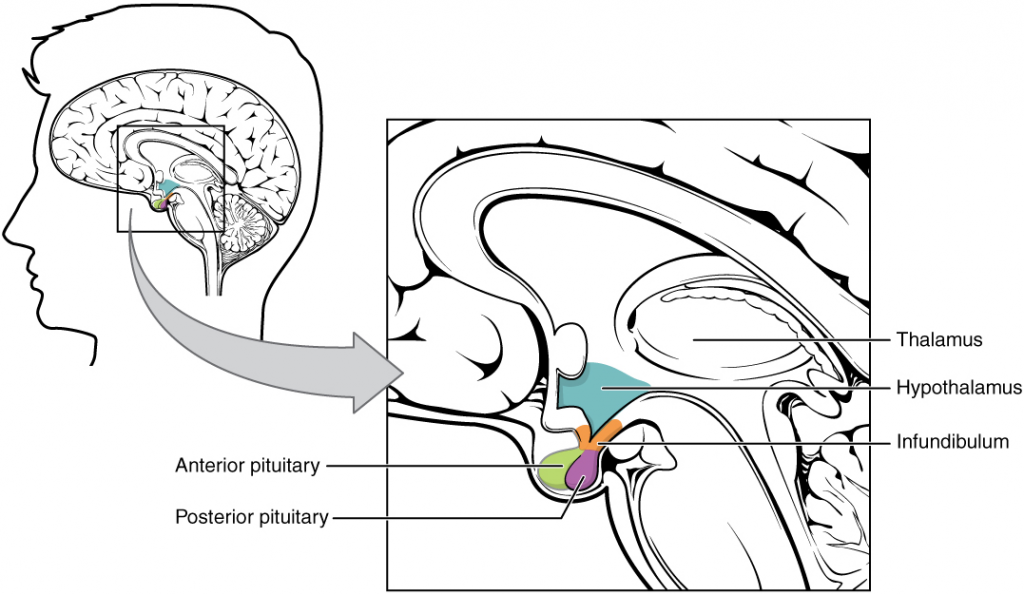
Table 14.3.3. Pituitary hormones
| Pituitary lobe | Associated hormones | Chemical class | Effect |
| Anterior | Growth hormone
(GH) |
Protein | Promotes growth of body tissues |
| Prolactin
(PRL) |
Peptide | Promotes milk production from mammary glands | |
| Thyroid-stimulating hormone
(TSH) |
Glycoprotein | Stimulates thyroid hormone release from thyroid | |
| Adrenocorticotropic hormone
(ACTH) |
Peptide | Stimulates hormone release by adrenal cortex | |
| Follicle-stimulating hormone
(FSH) |
Glycoprotein | Stimulates gamete production in gonads | |
| Luteinising hormone
(LH) |
Glycoprotein | Stimulates androgen production by gonads | |
| Posterior | Antidiuretic hormone
(ADH) |
Peptide | Stimulates water reabsorption by kidney |
| Oxytocin | Peptide | Stimulates uterine contractions during childbirth | |
| Intermediate zone | Melanocyte-stimulating hormone | Peptide | Simulates melanin formation in melanocytes |
Posterior Pituitary
The posterior pituitary is actually an extension of the neurons of the paraventricular and supraoptic nuclei of the hypothalamus. The cell bodies of these regions rest in the hypothalamus, but their axons descend as the hypothalamic–hypophyseal tract within the infundibulum, and end in axon terminals that comprise the posterior pituitary (14.3.2).
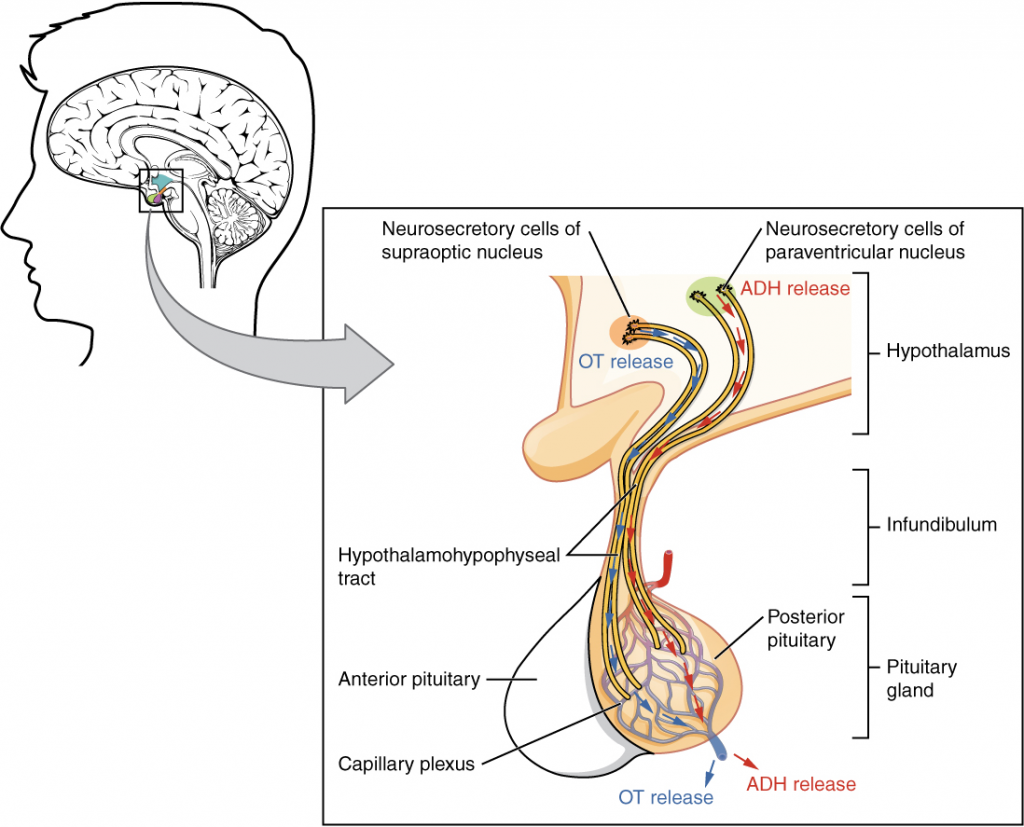
The posterior pituitary gland does not produce hormones, but rather stores and secretes hormones produced by the hypothalamus. The paraventricular nuclei produce the hormone oxytocin, whereas the supraoptic nuclei produce ADH. These hormones travel along the axons into storage sites in the axon terminals of the posterior pituitary. In response to signals from the same hypothalamic neurons, the hormones are released from the axon terminals into the bloodstream.
Oxytocin (OT)
When foetal development is complete, the peptide-derived hormone oxytocin (tocia- = “childbirth”) stimulates uterine contractions and dilation of the cervix. Throughout most of pregnancy, oxytocin hormone receptors are not expressed at high levels in the uterus. Toward the end of pregnancy, the synthesis of oxytocin receptors in the uterus increases, and the smooth muscle cells of the uterus become more sensitive to its effects. Oxytocin is continually released throughout childbirth through a positive feedback mechanism. As noted earlier, oxytocin prompts uterine contractions that push the foetal head toward the cervix. In response, cervical stretching stimulates additional oxytocin to be synthesised by the hypothalamus and released from the pituitary. This increases the intensity and effectiveness of uterine contractions and prompts additional dilation of the cervix. The feedback loop continues until birth.
Although the mother’s high blood concentrations of oxytocin begin to decrease immediately following birth, oxytocin continues to play a role in maternal and newborn health. First, oxytocin is necessary for the milk ejection reflex (commonly referred to as “let-down”) in breastfeeding women. As the newborn begins suckling, sensory receptors in the nipples transmit signals to the hypothalamus. In response, oxytocin is secreted and released into the bloodstream. Within seconds, cells in the mother’s milk ducts contract, ejecting milk into the infant’s mouth. Secondly, in both males and females, oxytocin is thought to contribute to parent–newborn bonding, known as attachment. Oxytocin is also thought to be involved in feelings of love and closeness, as well as in the sexual response.
Antidiuretic Hormone (ADH)
The solute concentration of the blood, or blood osmolarity, may change in response to the consumption of certain foods and fluids, as well as in response to disease, injury, medications, or other factors. Blood osmolarity is constantly monitored by osmoreceptors—specialised cells within the hypothalamus that are particularly sensitive to the concentration of sodium ions and other solutes.
In response to high blood osmolarity, which can occur during dehydration or following a very salty meal, the osmoreceptors signal the posterior pituitary to release antidiuretic hormone (ADH). The target cells of ADH are located in the tubular cells of the kidneys. Its effect is to increase epithelial permeability to water, allowing increased water reabsorption. The more water reabsorbed from the filtrate, the greater the amount of water that is returned to the blood and the less that is excreted in the urine. A greater concentration of water results in a reduced concentration of solutes. ADH is also known as vasopressin because, in very high concentrations, it causes constriction of blood vessels, which increases blood pressure by increasing peripheral resistance. The release of ADH is controlled by a negative feedback loop. As blood osmolarity decreases, the hypothalamic osmoreceptors sense the change and prompt a corresponding decrease in the secretion of ADH. As a result, less water is reabsorbed from the urine filtrate.
Interestingly, drugs can affect the secretion of ADH, for example, alcohol consumption inhibits the release of ADH, resulting in increased urine production that can eventually lead to dehydration and a hangover. A disease called diabetes insipidus is characterised by chronic underproduction of ADH that causes chronic dehydration. Because little ADH is produced and secreted, not enough water is reabsorbed by the kidneys. Although patients feel thirsty, and increase their fluid consumption, this doesn’t effectively decrease the solute concentration in their blood because ADH levels are not high enough to trigger water reabsorption in the kidneys. Electrolyte imbalances can occur in severe cases of diabetes insipidus.
Anterior Pituitary
The anterior pituitary originates from the digestive tract in the embryo and migrates toward the brain during foetal development. There are three regions: the pars distalis is the most anterior, the pars intermedia is adjacent to the posterior pituitary, and the pars tuberalis is a slender “tube” that wraps the infundibulum.
Recall that the posterior pituitary does not synthesise hormones, but merely stores them. In contrast, the anterior pituitary does manufacture hormones. However, the secretion of hormones from the anterior pituitary is regulated by two classes of hormones. These hormones—secreted by the hypothalamus—are the releasing hormones that stimulate the secretion of hormones from the anterior pituitary and the inhibiting hormones that inhibit secretion.
Hypothalamic hormones are secreted by neurons but enter the anterior pituitary through blood vessels (Figur 14.3.3). Within the infundibulum is a bridge of capillaries that connects the hypothalamus to the anterior pituitary. This network, called the hypophyseal portal system, allows hypothalamic hormones to be transported to the anterior pituitary without first entering the systemic circulation. The system originates from the superior hypophyseal artery, which branches off the carotid arteries and transports blood to the hypothalamus. The branches of the superior hypophyseal artery form the hypophyseal portal system (see Figure 14.3.3). Hypothalamic releasing and inhibiting hormones travel through a primary capillary plexus to the portal veins, which carry them into the anterior pituitary. Hormones produced by the anterior pituitary (in response to releasing hormones) enter a secondary capillary plexus, and from there drain into the circulation.
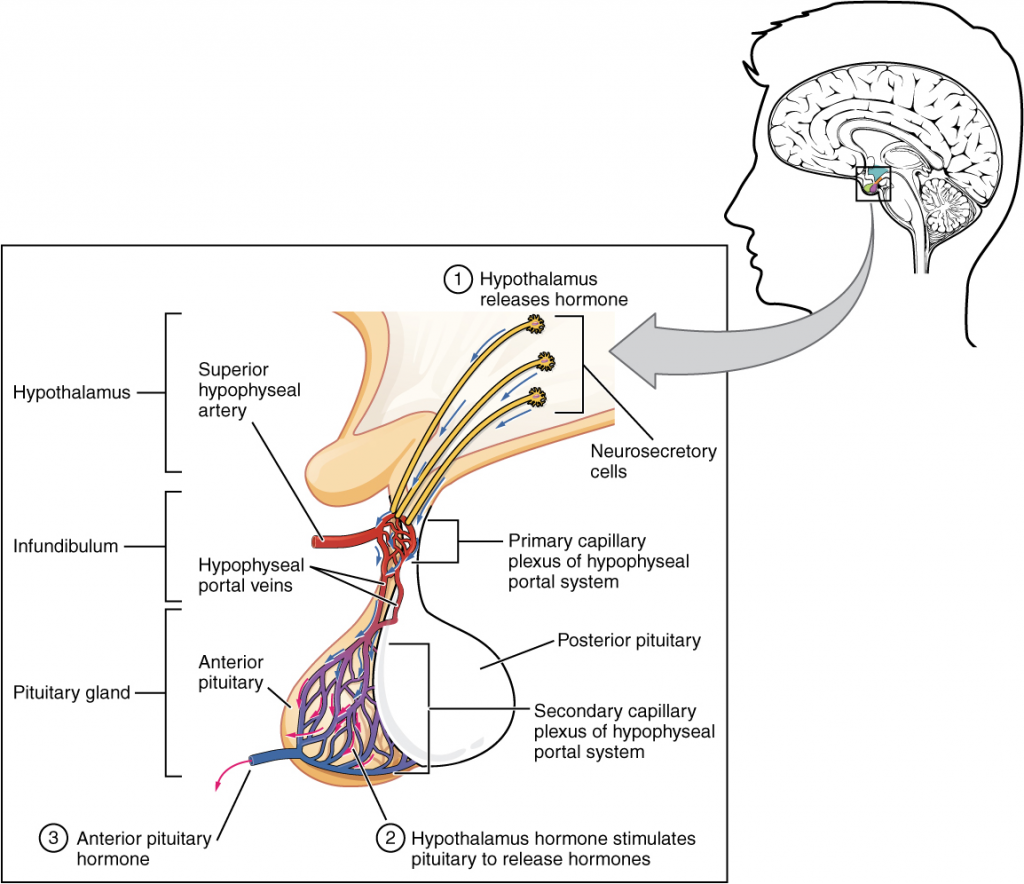
The anterior pituitary produces seven hormones. These are the growth hormone (GH), thyroid-stimulating hormone (TSH), adrenocorticotropic hormone (ACTH), follicle-stimulating hormone (FSH), luteinising hormone (LH), beta endorphin and prolactin. Of the hormones of the anterior pituitary, TSH, ACTH, FSH, and LH are collectively referred to as tropic hormones (trope- = “turning”) because they turn on or off the function of other endocrine glands.
Growth Hormone (GH)
The endocrine system regulates the growth of the human body, protein synthesis and cellular replication. A major hormone involved in this process is growth hormone (GH), also called somatotropin—a protein hormone produced and secreted by the anterior pituitary gland. Its primary function is anabolic; it promotes protein synthesis and tissue building through direct and indirect mechanisms (14.3.4). GH levels are controlled by the release of GHRH and GHIH (also known as somatostatin) from the hypothalamus.
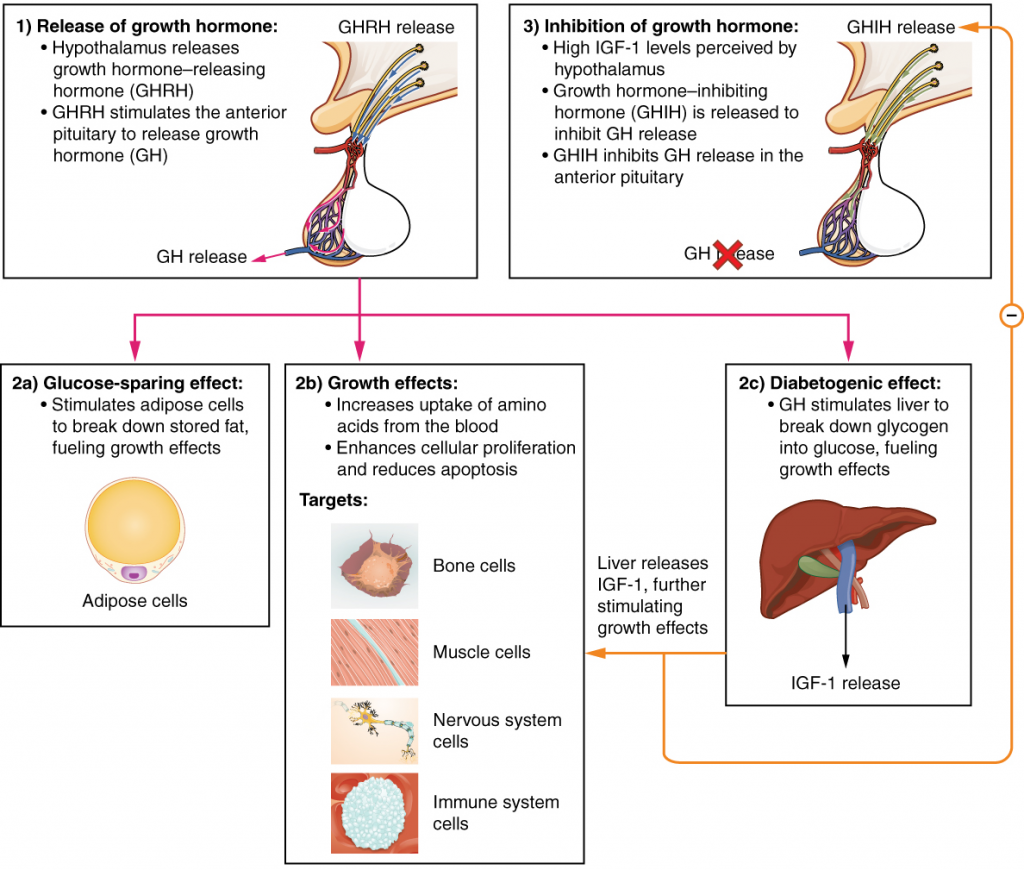
A glucose-sparing effect occurs when GH stimulates lipolysis, or the breakdown of adipose tissue, releasing fatty acids into the blood. As a result, many tissues switch from glucose to fatty acids as their main energy source, which means that less glucose is taken up from the bloodstream.
GH also initiates the diabetogenic effect in which GH stimulates the liver to break down glycogen to glucose, which is then deposited into the blood. The name “diabetogenic” is derived from the similarity in elevated blood glucose levels observed between individuals with untreated diabetes mellitus and individuals experiencing GH excess. Blood glucose concentrations rise as the result of a combination of glucose-sparing and diabetogenic effects.
GH indirectly mediates growth and protein synthesis by triggering the liver and other tissues to produce a group of proteins called insulin-like growth factors (IGFs). These proteins enhance cellular proliferation and inhibit apoptosis, or programmed cell death. IGFs stimulate cells to increase their uptake of amino acids from the blood for protein synthesis. Skeletal muscle and cartilage cells are particularly sensitive to stimulation from IGFs.
Dysfunction of the endocrine system’s control of growth can result in several disorders, for example, gigantism is a disorder in children that is caused by the secretion of abnormally large amounts of GH, resulting in excessive growth. A similar condition in adults is acromegaly, a disorder that results in the growth of bones in the face, hands, and feet in response to excessive levels of GH in individuals who have stopped growing. Abnormally low levels of GH in children can cause growth impairment—a disorder called pituitary dwarfism (also known as growth hormone deficiency).
Thyroid-Stimulating Hormone (TSH)
The activity of the thyroid gland is regulated by thyroid-stimulating hormone (TSH), also called thyrotropin. TSH is released from the anterior pituitary in response to thyrotropin-releasing hormone (TRH) from the hypothalamus. As discussed shortly, it triggers the secretion of thyroid hormones by the thyroid gland. In a classic negative feedback loop, elevated levels of thyroid hormones in the bloodstream then trigger a drop in production of TRH and subsequently TSH.
Adrenocorticotropic Hormone (ACTH)
The adrenocorticotropic hormone (ACTH), also called corticotropin, stimulates the adrenal cortex (the more superficial “bark” of the adrenal glands) to secrete corticosteroid hormones such as cortisol. ACTH come from a precursor molecule known as pro-opiomelanotropin (POMC) which produces several biologically active molecules when cleaved, including ACTH, melanocyte-stimulating hormone, and the brain opioid peptides known as endorphins.
The release of ACTH is regulated by the corticotropin-releasing hormone (CRH) from the hypothalamus in response to normal physiologic rhythms. A variety of stressors can also influence its release, and the role of ACTH in the stress response is discussed later in this chapter.
Follicle-Stimulating Hormone (FSH) and Luteinsing Hormone (LH)
The endocrine glands secrete a variety of hormones that control the development and regulation of the reproductive system (these glands include the anterior pituitary, the adrenal cortex, and the gonads—the testes in males and the ovaries in females). Much of the development of the reproductive system occurs during puberty and is marked by the development of sex-specific characteristics in both male and female adolescents. Puberty is initiated by gonadotropin-releasing hormone (GnRH), a hormone produced and secreted by the hypothalamus. GnRH stimulates the anterior pituitary to secrete gonadotropins—hormones that regulate the function of the gonads. The levels of GnRH are regulated through a negative feedback loop; high levels of reproductive hormones inhibit the release of GnRH. Throughout life, gonadotropins regulate reproductive function and, in the case of women, the onset and cessation of reproductive capacity.
The gonadotropins include two glycoprotein hormones: follicle-stimulating hormone (FSH) stimulates the production and maturation of sex cells, or gametes, including ova in women and sperm in men. FSH also promotes follicular growth; these follicles then release oestrogens in the female ovaries. Luteinising hormone (LH) triggers ovulation in women, as well as the production of oestrogens and progesterone by the ovaries. LH stimulates production of testosterone by the male testes.
Prolactin (PRL)
As its name implies, prolactin (PRL) promotes lactation (milk production) in women. During pregnancy, it contributes to development of the mammary glands, and after birth, it stimulates the mammary glands to produce breast milk. However, the effects of prolactin depend heavily upon the permissive effects of oestrogens, progesterone, and other hormones. And as noted earlier, the let-down of milk occurs in response to stimulation from oxytocin.
In a non-pregnant woman, prolactin secretion is inhibited by prolactin-inhibiting hormone (PIH), which is actually the neurotransmitter dopamine, and is released from neurons in the hypothalamus. Only during pregnancy do prolactin levels rise in response to prolactin-releasing hormone (PRH) from the hypothalamus.
Intermediate Pituitary: Melanocyte-Stimulating Hormone (MSH)
The cells in the zone between the pituitary lobes secrete a hormone known as melanocyte-stimulating hormone (MSH) that is formed by cleavage of the pro-opiomelanocortin (POMC) precursor protein. Local production of MSH in the skin is responsible for melanin production in response to UV light exposure. The role of MSH made by the pituitary is more complicated. For instance, people with lighter skin generally have the same amount of MSH as people with darker skin. Nevertheless, this hormone is capable of darkening of the skin by inducing melanin production in the skin’s melanocytes. Women also show increased MSH production during pregnancy; in combination with oestrogens, it can lead to darker skin pigmentation, especially the skin of the areolas and labia minora. Figure 14.3.5 is a summary of the pituitary hormones and their principal effects.
Section Review
The hypothalamus–pituitary complex is located in the diencephalon of the brain. The hypothalamus and the pituitary gland are connected by a structure called the infundibulum, which contains vasculature and nerve axons. The pituitary gland is divided into two distinct structures with different embryonic origins. The posterior lobe houses the axon terminals of hypothalamic neurons. It stores and releases into the bloodstream two hypothalamic hormones: oxytocin (OT) and antidiuretic hormone (ADH). The anterior lobe is connected to the hypothalamus by vasculature in the infundibulum and produces and secretes six hormones. Their secretion is regulated, however, by releasing and inhibiting hormones from the hypothalamus. The six anterior pituitary hormones are: growth hormone (GH), thyroid-stimulating hormone (TSH), adrenocorticotropic hormone (ACTH), follicle-stimulating hormone (FSH), luteinising hormone (LH) and prolactin (PRL).
Review Questions
Critical Thinking Questions
Click the drop down below to review the terms learned from this chapter.

